Energy Research
At UC Berkeley I was a member of the Computational Nanoscience Group under Dr. Jeffrey C. Grossman and Dr. Lucas K. Wagner, and worked on morphology optimization of excitonic photovoltaics. Below I present a summary of this research.
Excitonic Photovoltaics
A heterojunction is the interface between two solid-state materials that have unequal bandgaps. An example is the p-n junction used often in material science and circuitry (diodes), in which a p-type and n-type semiconductor are brought into contact and electrons "jump" from one to the other to create a current (if an external voltage source is supplied). Unlike ohmic resistive elements, diodes provide a very precise energy difference across the junction for each electron, so that in elements like an LED, a very particular wavelength of light is produced by the de-excitation of the electrons as they cross the boundary, dependent on the materials being used. An ohmic lamp, on the other hand, produces a broad range of spectral frequencies in the infrared and beyond, effectively wasting energy in non-visible wavelengths.
Bulk heterojunctions (many such boundaries in a system) are also used in solar cells. In the common variety, photons striking crystalline silicon free an electron from its bond, creating an electron-hole pair that can diffuse through the cell and be extracted at electrodes to provide current, and hence are a source of electricity. However, crystalline silicon is expensive and difficult to manufacture in bulk, and an alternative heterojunction material would be preferable. Much work is currently being done on amorphous silicon, which does not have to be specially prepared and cooled to create crystal structures. Another option (my research) is using organic compounds, which are cheap and easy to make. For organic heterojunctions, the two types of material are usually conjugated carbon molecules capable of harnessing light and creating excitons (bound electron-hole pairs), which can then separate and diffuse to the waiting electrodes.
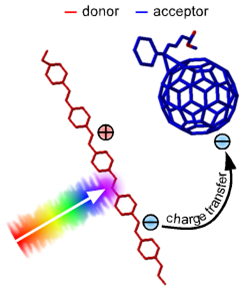
The molecule that absorbs light, creates the exciton, shuttles it around, and eventually transfers charge to the other molecule is called the donor. The receiver is called, plausibly enough, the acceptor. The donor is often a conjugated polymer such as the poly-3-hexylthiophene chain P3HT. The acceptor often used is the "buckyball," PCBM, and sometimes, both donor and acceptor can absorb photons and generate excitons. At the interface of the donor and acceptor, the exciton dissociates into an electron (transferred to the acceptor) and a hole (maintained on the donor). The free charges then wander throughout the regions in which they are bound, until they either are extracted to do something useful at the electrodes, or come close enough together to recombine.
The efficiency of the excitonic heterojunction is determined largely by the balance between transporting an exciton to the interface and transporting the resultant charges out of the cell without recombining (at an interface). The problem is largely geometric: how can we arrange the regions of donor and acceptor such that there are large enough conduction pathways for the free charges (so they won't annihilate one another), yet provide enough surface area of interface to dissociate excitons before they naturally decay (the lifetime of an exciton is less than a nanosecond, often much less). It is this question, one of morphology optimization, that can lead to large improvements in the sub-optimal efficiencies of organic solar cells being created today.
Our finalized paper can be seen here.
A presentation on organic photovoltaics can be found here.